Research
Research Interests:
Bioorganic Chemistry: amino acid metabolites, secondary metabolism (natural products chemistry), stereochemistry and mechanism of enzymatic reactions, organic synthesis with isotopes, new biosynthetic methods, NMR and structure elucidation of peptides and proteins; development of new medicinal agents.
The picture below shows a preferred conformation (three dimensional shape) of Carnocyclin A.
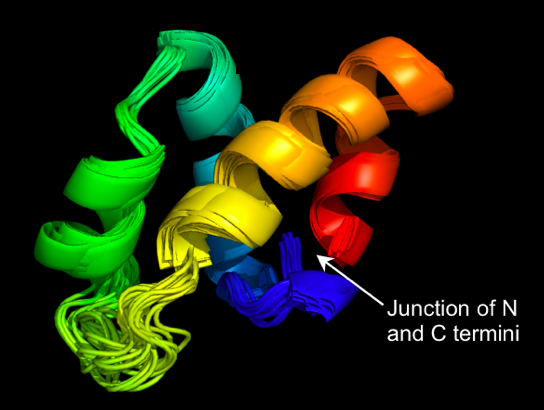
This is a cyclic non-toxic antimicrobial peptide that contains 60 amino acids and is produced naturally in meat products by Carnobacterium maltaromaticum UAL307, a lactic acid bacteria species that helps preserve food. This probiotic producing organism has been approved for commercial preservation of processed meat (e.g. sausages) in the USA and Canada. For a detailed description, see J. Biol. Chem. 284, 28674-28681 (2009).
Research Description:
Understanding the chemistry by which Nature assembles biological molecules is not only an exciting intellectual endeavour, but is also a prerequisite to rationally influence life processes in medicine and agriculture. Our research currently centers on the formation of important biological molecules, including antimicrobial peptides, amino acid metabolites, and polyketides. The approach is interdisciplinary. Experimental aspects of our projects encompass organic synthesis and spectroscopic methodology (especially NMR and mass spectrometry), as well as isotopic techniques, natural products isolation, enzymatic reactions, and culturing of microorganisms. Current projects include:
- Investigation of the three dimensional structure, mechanism of action, formation and applications of bacteriocins from lactic acid bacteria. These antimicrobial peptides (typically 30-80 amino acids) are non-toxic to mammals, naturally preserve food, and may be useful for treatment of gastrointestinal diseases.
- Examination of the mechanism of polyketide biosynthesis in fungi, especially formation of lovastatin (a widely-prescribed cholesterol-lowering drug) and generation of biologically active macrolides
- Construction of structurally modified neuropeptide hormones and their antagonists to provide improved activity and stability. These compounds influence a host of biological processes including lactation, childbirth, pain, appetite, pigmentation, pheromone biosynthesis and embryonic development.
- Understanding and duplicating the mechanisms of unusual enzymes, especially amino acid epimerases and hydroxylases (e.g. P450 and ketoglutarate dependent oxygenases).

|